
India Stem Cell Market Size, Share, Trends and Forecast by Product, Application, Technology, Therapy, End User, and Region, 2025-2033
India Stem Cell Market Overview:
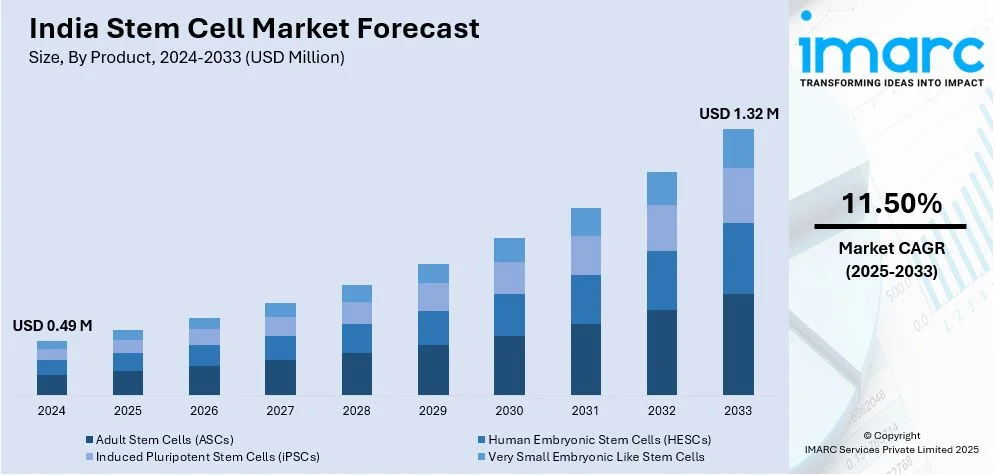
The India stem cell market size reached USD 0.49 Million in 2024. Looking forward, IMARC Group expects the market to reach USD 1.32 Million by 2033, exhibiting a growth rate (CAGR) of 11.50% during 2025-2033. The market is broadening with improving regenerative medicine, increasing biotech research investment, higher occurrences of chronic disease, and wider awareness of stem cell banking. Government support to biotechnology, need for targeted medicine, and partnerships between private companies and research institutions are further driving the growth of the market.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2024
|
|
Forecast Years
|
2025-2033
|
|
Historical Years
|
2019-2024
|
| Market Size in 2024 | USD 0.49 Million |
| Market Forecast in 2033 | USD 1.32 Million |
| Market Growth Rate (2025-2033) | 11.50% |
India Stem Cell Market Trends:
Expansion of Stem Cell Banking and Cord Blood Storage
Stem cell banking is witnessing immense traction in India as people become increasingly aware of its therapeutic possibilities in treating genetic diseases, blood disorders, and regenerative use. There is a demand surge for the storage of cord blood stem cells in response to parental interest in establishing enduring health safeguards for their children. Prominent market players are investing in advanced cryopreservation technology centers and expanding services. Government incentives in the form of regulations encouraging ethical stem cell banking and public-private partnerships are also fueling accessibility. Moreover, innovations in automated processing and storage technologies are improving the viability and potency of stored stem cells, making India a central hub for stem cell preservation in the Asia-Pacific region. For instance, in November 2023, CryoVault India received the "Best Stem Cell Bank in India" award from Telangana's Health Minister at the hmtv Healthcare Awards 2023.
To get more information on this market, Request Sample
Increasing Applications in Regenerative Medicine and Cell Therapy
Regenerative medicine is revolutionizing healthcare, with stem cells playing a critical role in the treatment of neurodegenerative disorders, cardiovascular diseases, orthopedic injuries, and diabetes. India is witnessing a surge in clinical trials and research focused on mesenchymal stem cells (MSCs) and induced pluripotent stem cells (iPSCs) for regenerative therapies. Hospitals and research institutes are partnering with biotechnology firms to accelerate commercialization. For instance, in February 2024, India conducted its first human clinical trial of gene therapy for haemophilia A at CMC Vellore, supported by the Department of Biotechnology and Emory University. The adoption of autologous stem cell therapies, where a patient’s own cells are used for treatment, is gaining traction due to minimal risk of rejection. With advancements in gene editing technologies such as CRISPR and improved differentiation protocols, the market is expected to see a broader range of therapeutic applications, attracting investment from domestic and international players.
Government and Private Sector Investments in Stem Cell Research
The Indian government is actively promoting stem cell research through funding programs, collaborations, and regulatory support. Initiatives like the Department of Biotechnology’s (DBT) National Biopharma Mission and investments from private biotech firms are accelerating research in stem cell-based therapies. India’s affordable clinical trial ecosystem, combined with a strong network of research institutions, is fostering innovation in cell-based treatments. Startups and multinational pharmaceutical companies are entering the market, leveraging India’s cost-effective research and skilled workforce. The development of GMP-certified (Good Manufacturing Practice) stem cell processing facilities is also increasing, ensuring compliance with global quality standards. These investments are expected to make India a leader in stem cell innovations, driving accessibility and affordability of therapies in the long run. For instance, in December 2024, Phase 2 clinical trials for CD19-directed CAR T-cell therapy were approved for children and adults with B-ALL and B-NHL. This therapy, developed by IIT Bombay and Tata Memorial Center, received Drug Controller General of India (DCGI) approval in March 2021 for Phase 1 trials, which have now been completed. The therapy has demonstrated safety and effectiveness, aligning with global standards. Research and development efforts continue with government support for expanding CAR T-cell applications in India.
India Stem Cell Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the region/country level for 2025-2033. Our report has categorized the market based on product, application, technology, therapy, and end user.
Product Insights:
- Adult Stem Cells (ASCs)
- Human Embryonic Stem Cells (HESCs)
- Induced Pluripotent Stem Cells (iPSCs)
- Very Small Embryonic Like Stem Cells
The report has provided a detailed breakup and analysis of the market based on the product. This includes adult stem cells (ASCs), human embryonic stem cells (HESCs), induced pluripotent stem cells (iPSCs), and very small embryonic like stem cells.
Application Insights:
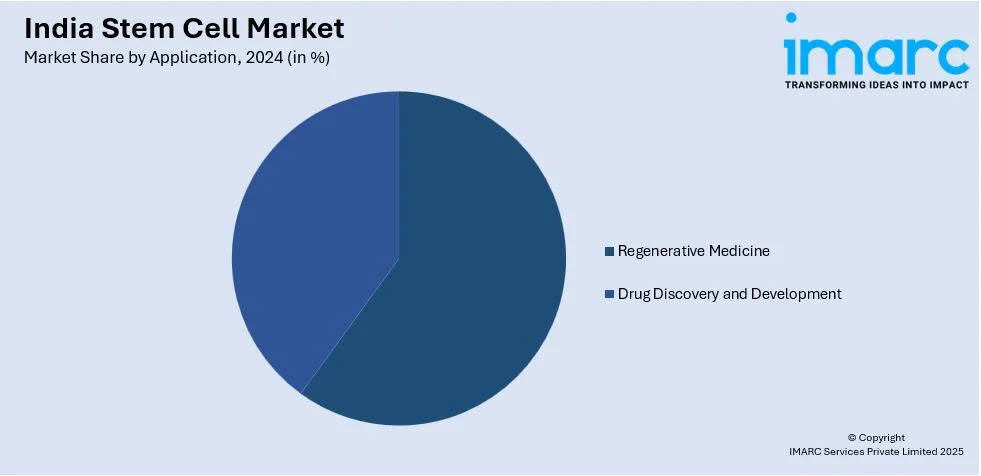
- Regenerative Medicine
- Drug Discovery and Development
A detailed breakup and analysis of the market based on the application have also been provided in the report. This includes regenerative medicine and drug discovery and development.
Technology Insights:
- Cell Acquisition
- Cell Production
- Cryopreservation
- Expansion and Sub-Culture.
A detailed breakup and analysis of the market based on the technology have also been provided in the report. This includes cell acquisition, cell production, cryopreservation, and expansion and sub-culture.
Therapy Insights:
- Autologous
- Allogeneic
A detailed breakup and analysis of the market based on the therapy have also been provided in the report. This includes autologous and allogeneic.
End User Insights:
- Pharmaceuticals and Biotechnology Companies
- Hospitals and Cell Banks
- Academic and Research Institutes
A detailed breakup and analysis of the market based on the end user have also been provided in the report. This includes pharmaceuticals and biotechnology companies, hospitals and cell banks, and academic and research institutes.
Regional Insights:
- North India
- South India
- East India
- West India
The report has also provided a comprehensive analysis of all the major regional markets, which include North, South, East, and West India.
Competitive Landscape:
The market research report has also provided a comprehensive analysis of the competitive landscape. Competitive analysis such as market structure, key player positioning, top winning strategies, competitive dashboard, and company evaluation quadrant has been covered in the report. Also, detailed profiles of all major companies have been provided.
India Stem Cell Market News:
- In July 2024, Bioserve India, a subsidiary of REPROCELL, launched advanced stem cell products in India, enhancing research in regenerative medicine and drug discovery. The product line includes cell culture solutions, reprogramming kits, 3D cell culture systems, and cellular assays. With a global biorepository of 600,000 biospecimens, the launch aims to accelerate stem cell research in India.
- In February 2024, Miltenyi Biotec launched its first India office in Hyderabad, investing in a Cell and Gene Therapy Center of Excellence (COE). The company will provide training and access to CGT technologies for Indian researchers and clinicians, from proof-of-concept to commercialization. Miltenyi aims to drive affordable CGT therapies in India, leveraging its CliniMACS Prodigy platform. The initiative highlights Miltenyi's commitment to fostering knowledge exchange and local manufacturing, with a focus on CAR T-cell therapy development.
India Stem Cell Market Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2024 |
| Historical Period | 2019-2024 |
| Forecast Period | 2025-2033 |
| Units | Million USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Products Covered | Adult Stem Cells (ASCs), Human Embryonic Stem Cells (HESCs), Induced Pluripotent Stem Cells (iPSCs), Very Small Embryonic Like Stem Cells |
| Applications Covered | Regenerative Medicine, Drug Discovery And Development |
| Technologies Covered | Cell Acquisition, Cell Production, Cryopreservation, Expansion and Sub-Culture |
| Therapies Covered | Autologous, Allogeneic |
| End Users Covered | Pharmaceuticals and Biotechnology Companies, Hospitals and Cell Banks, Academic and Research Institutes |
| Regions Covered | North India, South India, East India, West India |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the India stem cell market from 2019-2033.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the India stem cell market.
- Porter's Five Forces analysis assist stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the India stem cell industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Key Questions Answered in This Report
The stem cell market in India was valued at USD 0.49 Million in 2024.
The India stem cell market is projected to exhibit a CAGR of 11.50% during 2025-2033, reaching a value of USD 1.32 Million by 2033.
India stem cell market is expanding steadily, characterized by growing demand for regenerative and personalized therapies. Enhanced research infrastructure, rising chronic disease prevalence, and increased patient awareness are also fueling adoption. Collaboration between biotech firms, academic institutions, and healthcare providers is encouraging innovation and access to advanced stem cell treatments, further strengthening the market.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












