
India Orphan Drugs Market Size, Share, Trends and Forecast by Drug Type, Disease Type, Phase, Top Selling Drugs, Distribution Channel, and Region, 2025-2033
India Orphan Drugs Market Size and Share:
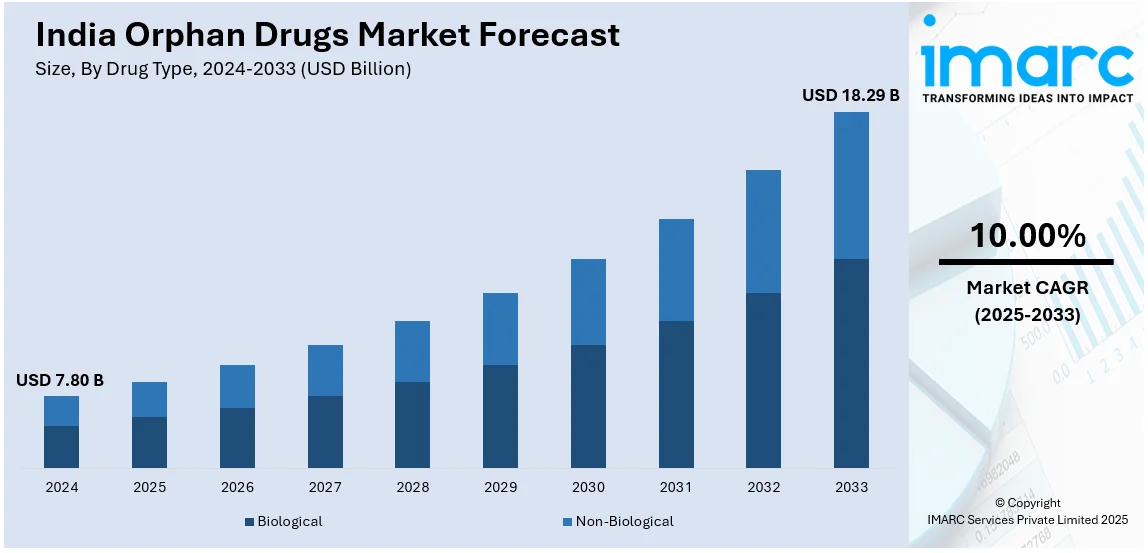
The India orphan drugs market size reached USD 7.80 Billion in 2024. Looking forward, IMARC Group expects the market to reach USD 18.29 Billion by 2033, exhibiting a growth rate (CAGR) of 10.00% during 2025-2033. The market is stimulated by increased rare disease awareness, government policy incentives, enhanced diagnostic capabilities, and patient advocacy. Progress in biotechnology, partnering among pharmaceutical companies, and incentives for the development of orphan drugs also drive market growth, satisfying unmet medical needs and treating more rare diseases.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2024
|
|
Forecast Years
|
2025-2033
|
|
Historical Years
|
2019-2024
|
| Market Size in 2024 | USD 7.80 Billion |
| Market Forecast in 2033 | USD 18.29 Billion |
| Market Growth Rate (2025-2033) | 10.00% |
India Orphan Drugs Market Trends:
Growing Government Support and Regulatory Reforms
India is building support for orphan drug development with regulatory reforms and incentives. The National Policy for Rare Diseases (NPRD) and health authorities' recognition of rare diseases have provided a conducive environment. Incentives like fast-track approvals, fee exemptions, and longer market exclusivity are stimulating pharmaceutical investments. The Central Drugs Standard Control Organization (CDSCO) is streamlining clinical trials and drug approvals to facilitate market entry. With India's patient population of rare diseases estimated at 70 to 96 million, the demand for orphan drugs is imperative. These policy reforms are driving innovation, enhancing access to treatment, and luring international pharmaceutical firms. Consequently, India's orphan drug industry is growing, providing new opportunities for pharmaceutical companies while filling the unmet medical needs of millions.
To get more information on this market, Request Sample
Rise in Public-Private Collaborations and Global Partnerships
One of the key trends in the Indian orphan drug market is growing alliances between public agencies, private pharmaceutical companies, and international biotech firms. Indian pharmaceutical companies are forming strategic partnerships with international companies for technology transfer, co-development, and marketing of orphan drugs. This trend narrows gaps in infrastructure, knowledge, and finance, speeding up drug development and market entry. Moreover, public-private partnerships with academic institutions and healthcare organizations are fostering research and innovation in rare disease therapies. Such collaborative efforts are critical to overcoming financial and technical challenges in orphan drug development while also bringing advanced treatment options to Indian patients through shared resources and global best practices.
Advancements in Genomic Research and Personalized Medicine
India’s progress in genomic research and precision medicine is strengthening its orphan drugs market. The Genome India Project (GIP), launched in 2020, has sequenced 10,000 genomes from diverse ethnic groups, advancing rare disease research. Investment in genetic diagnosis, bioinformatics, and molecular profiling is propelling targeted therapy and personalized care. Startups and research organizations are creating gene-based solutions and biomarker-led treatments specific to rare disease patients. As genetic sequencing costs decline and awareness improves, early diagnosis rates are rising, increasing demand for orphan drugs. The Indian Council of Medical Research’s National Registry for Rare Diseases, tracking over 15,000 cases, supports this shift. Precision medicine is transforming traditional treatment approaches, enabling individualized care and expanding orphan drug accessibility in India.
India Orphan Drugs Market Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the market, along with forecasts at the region level for 2025-2033 Our report has categorized the market based on drug type, disease type, phase, top selling drugs, and distribution channel.
Drug Type Insights:
- Biological
- Non-Biological
The report has provided a detailed breakup and analysis of the market based on the drug type. This includes biological, and non-biological.
Disease Type Insights:
- Oncology
- Hematology
- Neurology
- Cardiovascular
- Others
A detailed breakup and analysis of the market based on the disease type have also been provided in the report. This includes oncology, hematology, neurology, cardiovascular, and others.
Phase Insights:
- Phase I
- Phase II
- Phase III
- Phase IV
The report has provided a detailed breakup and analysis of the market based on the phase. This includes phase I, phase II, phase III, and phase IV.
Top Selling Drugs Insights:
- Revlimid
- Rituxan
- Copaxone
- Opdivo
- Keytruda
- Imbruvica
- Avonex
- Sensipar
- Soliris
- Others
A detailed breakup and analysis of the market based on the top selling drug have also been provided in the report. This includes Revlimid, Rituxan, Copaxone, Opdivo, Keytruda, Imbruvica, Avonex, Sensipar, Soliris, and others.
Distribution Channel Insights:
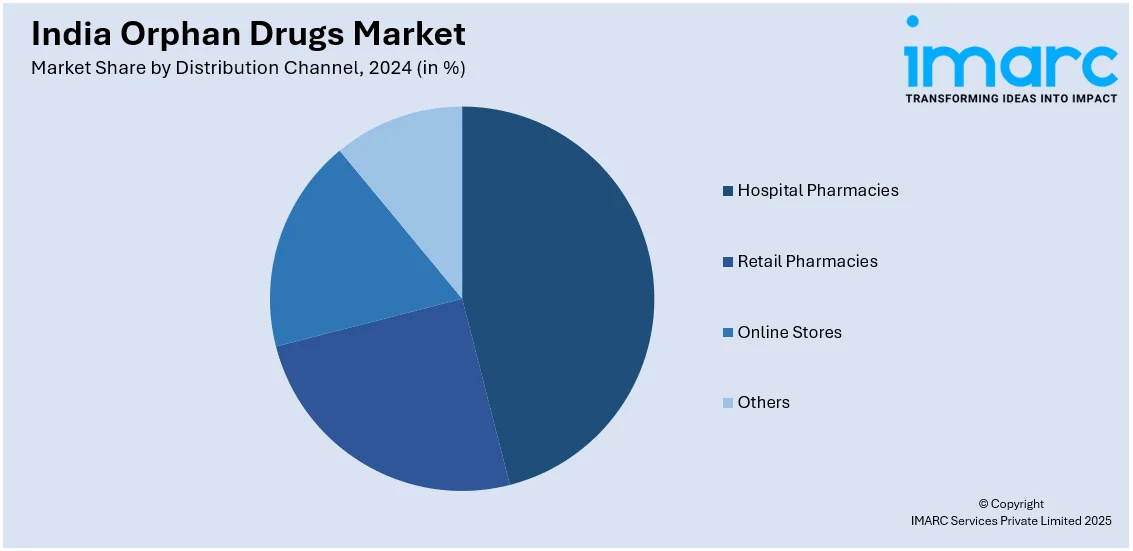
- Hospital Pharmacies
- Retail Pharmacies
- Online Stores
- Others
The report has provided a detailed breakup and analysis of the market based on the distribution channel. This includes hospital pharmacies, retail pharmacies, online stores, and others.
Regional Insights:
- North India
- South India
- East India
- West India
The report has also provided a comprehensive analysis of all the major regional markets, which include North, South, East, and West India
Competitive Landscape:
The market research report has also provided a comprehensive analysis of the competitive landscape. Competitive analysis such as market structure, key player positioning, top winning strategies, competitive dashboard, and company evaluation quadrant has been covered in the report. Also, detailed profiles of all major companies have been provided.
India Orphan Drugs Market News:
- In May 2024, Zydus Lifesciences’ US-based subsidiary, Sentynl Therapeutics Inc, acquired the global proprietary rights for Zokinvy, the only USFDA-approved treatment for progeria. Zokinvy is designed to address both the symptoms and underlying cause of the rare genetic disorder in patients aged over 12 months. This strategic move strengthens Zydus’ rare disease portfolio and marks a significant step in expanding access to advanced treatments for progeria worldwide.
India Orphan Drugs Market Report Coverage:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2024 |
| Historical Period | 2019-2024 |
| Forecast Period | 2025-2033 |
| Units | Billion USD |
| Scope of the Report |
Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Future Market Assessment:
|
| Drug Types Covered | Biological, Non-Biological |
| Disease Types Covered | Oncology, Hematology, Neurology, Cardiovascular, Others |
| Phases Covered | Phase I, Phase II, Phase III, Phase IV |
| Top Selling Drugs Covered | Revlimid, Rituxan, Copaxone, Opdivo, Keytruda, Imbruvica, Avonex, Sensipar, Soliris, Others |
| Distribution Channels Covered | Hospital Pharmacies, Retail Pharmacies, Online Stores, Others |
| Regions Covered | North India, South India, East India, West India |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC’s industry report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the India orphan drugs market from 2019-2033.
- The research report provides the latest information on the market drivers, challenges, and opportunities in the India orphan drugs market.
- Porter's five forces analysis assist stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the India orphan drugs industry and its attractiveness.
- Competitive landscape allows stakeholders to understand their competitive environment and provides an insight into the current positions of key players in the market.
Key Questions Answered in This Report
The orphan drugs market in India was valued at USD 7.80 Billion in 2024.
The India orphan drugs market is projected to exhibit a CAGR of 10.00% during 2025-2033, reaching a value of USD 18.29 Billion by 2033.
India’s orphan drugs market is driven by supportive government policies, growing awareness of rare diseases, improved diagnostic capabilities, and collaborations between pharmaceutical companies and healthcare providers. Increasing research in genomics and a focus on addressing unmet medical needs further fuel the development and availability of treatments for rare conditions.
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












