
Breast Cancer Liquid Biopsy Market Report by Product Service (Reagent Kits, Instruments, Services), Circulating Biomaker (Circulating Tumor Cells (CTCs), Cell-Free DNA (cfDNA), Extracelluar Vesicles (EVs), and Others), End User (Reference Laboratories, Hospital and Physician Laboratories, and Other), and Region 2026-2034
Global Breast Cancer Liquid Biopsy Market:
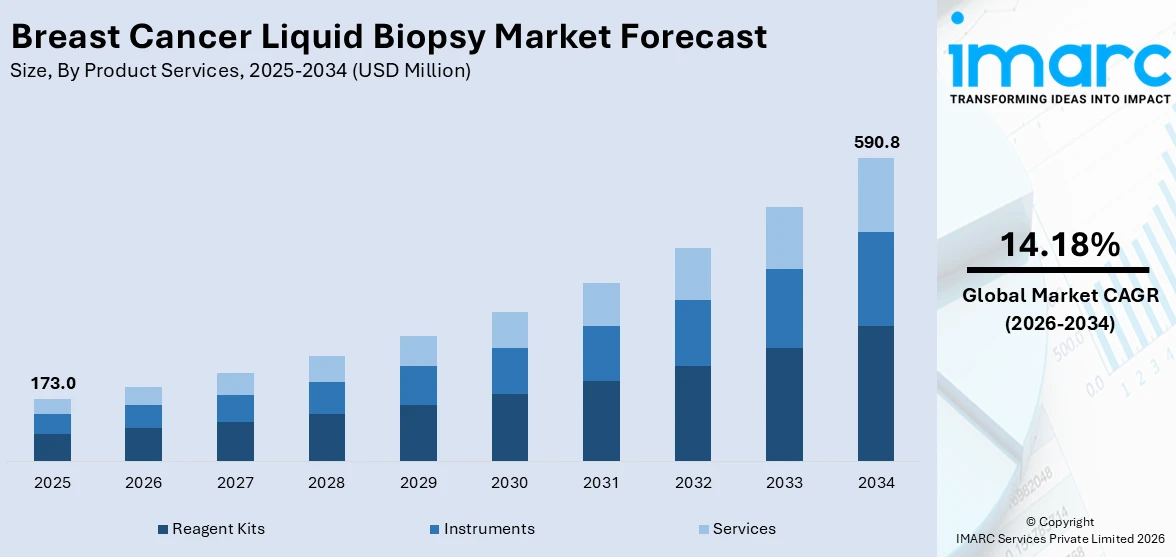
The global breast cancer liquid biopsy market size reached USD 173.0 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 590.8 Million by 2034, exhibiting a growth rate (CAGR) of 14.18% during 2026-2034. North America dominates the market, driven by the ongoing shift towards non-invasive diagnostic methods and supportive government initiatives for cancer screening. The rising prevalence of breast cancer, technological advancements, and surging investments in cancer research are primarily fueling the market growth.
|
Report Attribute
|
Key Statistics
|
|---|---|
|
Base Year
|
2025
|
|
Forecast Years
|
2026-2034
|
|
Historical Years
|
2020-2025
|
|
Market Size in 2025
|
USD 173.0 Million |
|
Market Forecast in 2034
|
USD 590.8 Million |
| Market Growth Rate 2026-2034 | 14.18% |
Rapid technological advancements, such as digital polymerase chain reaction (PCR), next-generation sequencing (NGS), and artificial intelligence (AI)-based data analytics, have significantly improved the sensitivity, accuracy, and clinical utility of liquid biopsy tests. As circulating tumor DNA (ctDNA) and circulating tumor cells (CTCs) are employed not only for early detection but also for monitoring treatment response, identifying minimal residual illness, and predicting relapse, their growing clinical applications are also contributing to the market share. Product developments and clinical validation are being accelerated by the increasing investments made in oncology diagnostics research by both public and private organizations. Moreover, liquid biopsy is gaining traction as a companion diagnostic tool due to the heightened focus on targeted therapeutics.
To get more information on this market Request Sample
Breast Cancer Liquid Biopsy Market Trends:
Rising prevalence of breast cancer cases
Increasing incidence of breast cancer cases is one of the key factors fueling the growth of the market, as it is creating an urgent requirement for effective diagnostic and monitoring tools. As per the National Breast Cancer Foundation, in the US, it is projected that by 2025, 316,950 women will be diagnosed with invasive breast cancer, along with 59,080 new diagnoses of non-invasive (in situ) breast cancer. As more women are being diagnosed each year, the demand for non-invasive, precise, and reliable techniques for tumor detection and disease monitoring is growing. Liquid biopsy enables the identification of tumor-derived biomarkers in blood samples, offering a safer and faster option compared to conventional tissue biopsies. The increasing burden of breast cancer underscores the significance of early detection and continuous monitoring to enhance survival rates and treatment results, positioning liquid biopsy as an essential solution that is experiencing widespread acceptance globally.
Increasing focus on personalized and precision medicine
The growing emphasis on personalized and precision medicine is positively influencing the market, as this approach requires advanced tools to identify individual tumor characteristics and guide targeted therapies. As per the IMARC Group, the global precision medicine market size reached USD 82.1 Billion in 2024. Liquid biopsy enables real-time tracking of genetic mutations, tumor heterogeneity, and resistance mechanisms, allowing oncologists to tailor treatments according to a patient’s unique cancer profile. This capability enhances treatment effectiveness, reduces unnecessary side effects, and ensures better patient compliance. As breast cancer presents varied molecular subtypes, precision diagnostics like liquid biopsy are becoming indispensable in delivering optimal therapeutic strategies. With healthcare systems focusing on precision medicine, the integration of liquid biopsy into clinical practice is accelerating.
Supportive government initiatives for cancer screening
Supportive government initiatives for cancer screening are offering a favorable market outlook. In February 2025, the health department of Kerala launched a large-scale cancer screening program, ‘Arogyam Anandam - Keep Cancer at Bay,’ as a part of a year-long campaign aimed at early detection of cancer and ensuring prompt treatment. In this timeframe, 855 governmental health centers, certain private hospitals, and special camps would offer cancer screenings, which encompassed basic breast cancer check-ups and Pap smear examinations for cervical cancer. Many national health programs are prioritizing early detection and preventive care, leading to rising adoption of advanced diagnostic methods. Liquid biopsy, being minimally invasive (MI) and cost-effective compared to surgical biopsies, aligns well with public health goals to expand access to screening and monitoring. Additionally, awareness campaigns run by government agencies and public health bodies are increasing the acceptance of novel technologies like liquid biopsy among both patients and clinicians.
Key Growth Drivers of Breast Cancer Liquid Biopsy Market:
Technological advancements
Technological advancements are propelling the market growth. Innovations in molecular diagnostics, genomic sequencing, and biomarker analysis aid in enhancing the accuracy and clinical utility of liquid biopsy tests. Cutting-edge platforms enable the detection of minute levels of circulating tumor cells (CTCs), providing earlier and more reliable insights into cancer progression and treatment response. Advancements in NGS and digital PCR technologies have improved sensitivity and reduced turnaround time, making liquid biopsy increasingly practical in routine oncology care. Moreover, integration with AI and bioinformatics allows deeper analysis of complex datasets, further refining predictive capabilities. These innovations are not only expanding clinical applications but also increasing physician confidence, thereby driving widespread adoption and supporting strong market growth.
Rising investments in oncology diagnostics research
High spending on oncology diagnostics research is playing a crucial role in fueling the market growth by accelerating product development. Both public and private sectors are channeling funds into research activities to enhance cancer detection, monitoring, and treatment personalization. This surge in investment is supporting clinical trials, validation studies, and the commercialization of advanced assays, making liquid biopsy more accessible and reliable for clinicians and patients. Increased venture capital interest and collaborations between biotech firms, diagnostic companies, and research institutions are also fueling rapid advancements. These investments not only lead to technological improvements but also help lower costs and expand availability, thereby strengthening adoption rates. Consequently, robust financial support for oncology research is boosting the expansion of the market.
Expanding clinical applications of ctDNA
Increasing clinical applications of ctDNA are bolstering the market growth, as ctDNA provides critical insights into tumor biology and disease progression. Beyond early diagnosis, ctDNA analysis is increasingly being used to monitor treatment effectiveness, detect minimal residual disease (MRD), and identify potential relapse before clinical symptoms appear. This real-time monitoring capability allows oncologists to adjust therapies promptly, improving patient outcomes and survival rates. Additionally, ctDNA enables the detection of specific genetic mutations and resistance mechanisms, supporting the growing trend of targeted therapies and personalized medicine. As ctDNA applications are expanding into prognosis, recurrence monitoring, and drug development, healthcare providers are incorporating liquid biopsy into clinical workflows. This broadening of clinical utility is driving higher adoption, thereby stimulating the market growth on a global scale.
Global Breast Cancer Liquid Biopsy Industry Segmentation:
IMARC Group provides an analysis of the key trends in each segment of the global breast cancer liquid biopsy market report, along with forecasts at the global, regional, and country levels from 2026-2034. Our report has categorized the market based on product services, circulating biomarker, and end user.
Breakup by Product Services:
- Reagent Kits
- Instruments
- Services
Currently, reagent kits account for the majority of the global market share
The report has provided a detailed breakup and analysis of the breast cancer liquid biopsy market based on the product services. This includes reagent kits, instruments, and services. According to the report, reagent kits account for the majority of the global market share.
According to the breast cancer liquid biopsy market outlook, the increasing global incidence of breast cancer has heightened the need for better diagnostic, monitoring, and screening tools. As liquid biopsy becomes more commonly used, the demand for reagent kits that support these tests rises proportionally. Moreover, traditional tissue biopsies are invasive and can cause discomfort and complications. Liquid biopsy offers a non-invasive alternative, which is gaining popularity for both diagnostic and treatment monitoring purposes. This shift is a key driver for reagent kits used in liquid biopsy procedures, such as those for detecting circulating tumor DNA (ctDNA) or circulating tumor cells (CTCs).
Breakup by Circulating Biomarker:
- Circulating Tumor Cells (CTCs)
- Cell-Free DNA (cfDNA)
- Extracellular Vesicles (EVs)
- Others
The report has provided a detailed breakup and analysis of the breast cancer liquid biopsy market based on the circulating biomarker. This includes circulating tumor cells (CTCs), cell-free DNA (cfDNA), extracellular vesicles (EVs), and others.
According to the breast cancer liquid biopsy market analysis report, CTCs are cancer cells that have shed from the primary tumor into the bloodstream. The presence of CTCs in the blood correlates with disease stage and prognosis. Higher numbers of CTCs are often associated with advanced cancer and poorer outcomes. Moreover, cfDNA consists of small fragments of DNA released from tumor cells (also referred to as circulating tumor DNA or ctDNA) and other cells into the bloodstream. Moreover, analysis of cfDNA allows for the detection of specific mutations, such as those in the PIK3CA gene, TP53, or ESR1, which can guide treatment choices, especially for targeted therapies. Furthermore, extracellular vesicles (such as exosomes and microvesicles) are small membrane-bound particles released by cells, including cancer cells, into the bloodstream. EVs contain a variety of biomolecules, including proteins, RNA, DNA, and lipids, and can play a role in cell-to-cell communication.
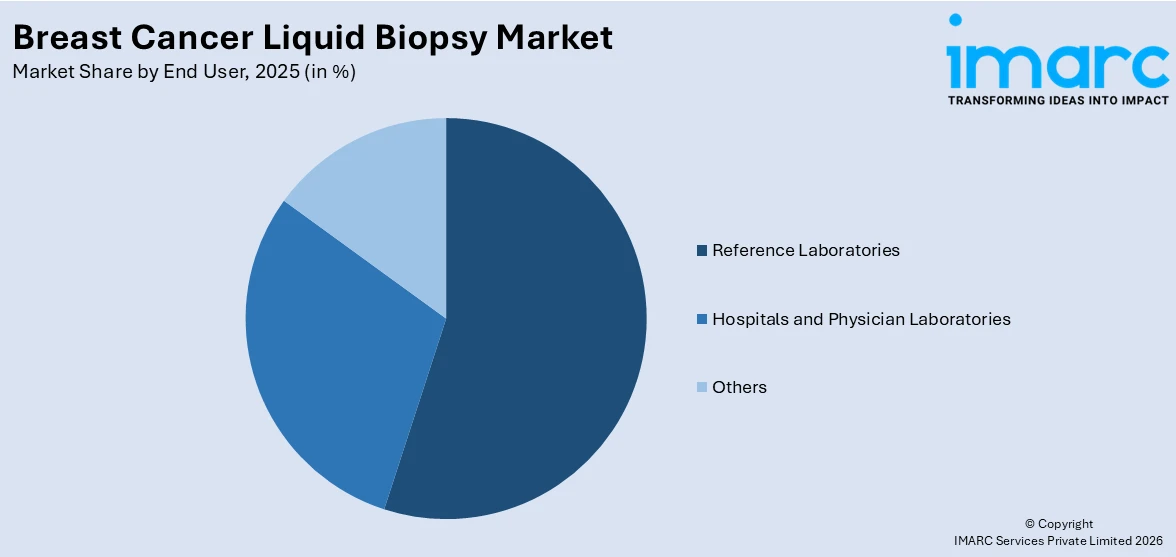
Breakup by End User:
Access the comprehensive market breakdown Request Sample
- Reference Laboratories
- Hospitals and Physician Laboratories
- Others
The report has provided a detailed breakup and analysis of the breast cancer liquid biopsy market based on the end user. This includes reference laboratories, hospitals and physician laboratories, and others.
According to the breast cancer liquid biopsy market outlook report, reference labs often have access to the most cutting-edge technologies in liquid biopsy, including next-generation sequencing (NGS) and circulating tumor DNA (ctDNA) analysis. Their advanced infrastructure attracts demand from hospitals and physician labs that do not have these capabilities. Moreover, large hospitals, especially those with dedicated oncology centers, have a high volume of breast cancer patients requiring regular monitoring. Liquid biopsy offers a less invasive, faster alternative to traditional tissue biopsies for diagnosis, treatment monitoring, and recurrence detection. Furthermore, physical labs are often the first point of care for breast cancer screening. Liquid biopsies provide a more accessible and less invasive option for patients at high risk of breast cancer, allowing for earlier detection and intervention.
Breakup by Region:
- North America
- United States
- Canada
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Others
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Russia
- Others
- Latin America
- Brazil
- Mexico
- Others
- Middle East and Africa
North America currently dominates the global market
The report has also provided a comprehensive analysis of all the major regional markets, which include North America (the United States and Canada); Europe (Germany, France, the United Kingdom, Italy, Spain, Russia, and others); Asia Pacific (China, Japan, India, South Korea, Australia, Indonesia, and others); Latin America (Brazil, Mexico, and others); and the Middle East and Africa. According to the report, North America dominates the global market.
According to the breast cancer liquid biopsy market statistics, breast cancer remains one of the most common cancers among women in North America, driving demand for improved diagnostic tools. Moreover, liquid biopsies offer a non-invasive alternative to traditional tissue biopsies, which can be painful, risky, and difficult to perform, especially in advanced cancer cases. As a simple blood test, liquid biopsy is more appealing to patients, contributing to its growing adoption. Besides this, advances in liquid biopsy technologies, including next-generation sequencing (NGS), PCR-based methods, and improved ctDNA and CTC (circulating tumor cells) detection platforms, have significantly increased the accuracy and sensitivity of these tests. These innovations are driving wider clinical adoption of liquid biopsy for breast cancer diagnosis and monitoring.
Competitive Landscape:
The breast cancer liquid biopsy market research report has provided a comprehensive analysis of the competitive landscape. Detailed profiles of all major market companies have also been provided. Some of the key players in the market include:
- Biocept Inc.
- Bio-Rad Laboratories Inc.
- F. Hoffmann-La Roche AG
- Fluxion Biosciences Inc.
- Guardant Health Inc.
- Illumina Inc.
- Menarini Silicon Biosystems (The Menarini Group)
- NeoGenomics Laboratories Inc.
- Qiagen
- Sysmex Europe SE (Sysmex Corporation)
(Please note that this is only a partial list of the key players, and the complete list is provided in the report.)
Breast Cancer Liquid Biopsy Market News:
- June 2025: A start-up backed by the C3N-IA Science Park at Universidad Carlos III de Madrid (UC3M) and focused on oncology, created a tool to assess treatment response in solid tumor patients using a simple blood sample. The researchers managed to predict the occurrence of clinical relapses up to 68 months before symptoms recognizable by conventional methods emerged. The technique employed to identify relapses promptly started with an initial liquid biopsy. This method had potential for breast cancer, lung cancer, and blood cancers.
- June 2025: The study, released in the New England Journal of Medicine, examined individuals whose breast cancers were driven by estrogen. The research revealed that liquid biopsies could identify mutations as much as nine months prior to when the alterations would be visible on imaging scans. This allowed individuals to cease ineffective therapies as soon as possible and transition to those with improved prospects for managing the cancer.
- February 2025: The study, released in Precision Clinical Medicine, emphasized the potential of ctDNA analysis, a crucial element utilized in liquid biopsies, as a non-invasive approach to detect actionable biomarkers, allowing precision medicine to customize treatments for individual breast cancer patients. Through providing real-time genetic information, ctDNA testing could enhance patient results and decrease treatment resistance, facilitating more tailored care in oncology.
- September 2024: Strand Life Sciences, a Reliance Industries subsidiary, released the Somatic Advantage 74 Liquid Biopsy (SA74 LB) Test, which could identify ctDNA in cancer patients’ blood.
Breast Cancer Liquid Biopsy Market Report Scope:
| Report Features | Details |
|---|---|
| Base Year of the Analysis | 2025 |
| Historical Period | 2020-2025 |
| Forecast Period | 2026-2034 |
| Units | Million USD |
| Scope of the Report | Exploration of Historical Trends and Market Outlook, Industry Catalysts and Challenges, Segment-Wise Historical and Predictive Market Assessment:
|
| Product Services Covered | Reagent Kits, Instruments, Services |
| Circulating Biomarkers Covered | Circulating Tumor Cells (CTCs), Cell-Free DNA (cfDNA), Extracellular Vesicles (EVs), Others |
| End Users Covered | Reference Laboratories, Hospitals and Physician Laboratories, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Russia, China, Japan, India, South Korea, Australia, Indonesia, Brazil, Mexico |
| Companies Covered | Biocept Inc., Bio-Rad Laboratories Inc., F. Hoffmann-La Roche AG, Fluxion Biosciences Inc., Guardant Health Inc., Illumina Inc., Menarini Silicon Biosystems (The Menarini Group), NeoGenomics Laboratories Inc., Qiagen, Sysmex Europe SE (Sysmex Corporation), etc. |
| Customization Scope | 10% Free Customization |
| Post-Sale Analyst Support | 10-12 Weeks |
| Delivery Format | PDF and Excel through Email (We can also provide the editable version of the report in PPT/Word format on special request) |
Key Benefits for Stakeholders:
- IMARC's report offers a comprehensive quantitative analysis of various market segments, historical and current market trends, market forecasts, and dynamics of the breast cancer liquid biopsy market from 2020-2034.
- The research study provides the latest information on the market drivers, challenges, and opportunities in the global breast cancer liquid biopsy market.
- The study maps the leading, as well as the fastest-growing, regional markets. It further enables stakeholders to identify the key country-level markets within each region.
- Porter's five forces analysis assists stakeholders in assessing the impact of new entrants, competitive rivalry, supplier power, buyer power, and the threat of substitution. It helps stakeholders to analyze the level of competition within the breast cancer liquid biopsy industry and its attractiveness.
- The competitive landscape allows stakeholders to understand their competitive environment and provides insight into the current positions of key players in the market.
Frequently Asked Questions About the Breast Cancer Liquid Biopsy Market Report
The global breast cancer liquid biopsy market was valued at USD 173.0 Million in 2025.
We expect the global breast cancer liquid biopsy market to exhibit a CAGR of 14.18% during 2026-2034.
The sudden outbreak of the COVID-19 pandemic had led to postponement of elective breast cancer liquid biopsy to reduce the risk of the coronavirus infection upon hospital visits and interaction with healthcare professionals or medical equipment.
The rising prevalence of breast cancer, along with the increasing consumer awareness towards the numerous benefits of liquid biopsy techniques, such as quicker results, lower cost, minimal health risk, non-invasiveness, etc., is primarily driving the global breast cancer liquid biopsy market.
Based on the product services, the global breast cancer liquid biopsy market can be bifurcated into reagent kits, instruments, and services. Currently, reagent kits account for the majority of the global market share.
On a regional level, the market has been classified into North America, Asia-Pacific, Europe, Latin America, and Middle East and Africa, where North America currently dominates the global market.
Some of the major players in the global breast cancer liquid biopsy market include Biocept Inc., Bio-Rad Laboratories Inc., F. Hoffmann-La Roche AG, Fluxion Biosciences Inc., Guardant Health Inc., Illumina Inc., Menarini Silicon Biosystems (The Menarini Group), NeoGenomics Laboratories Inc., Qiagen, and Sysmex Europe SE (Sysmex Corporation).
Need more help?
- Speak to our experienced analysts for insights on the current market scenarios.
- Include additional segments and countries to customize the report as per your requirement.
- Gain an unparalleled competitive advantage in your domain by understanding how to utilize the report and positively impacting your operations and revenue.
- For further assistance, please connect with our analysts.
 Request Customization
Request Customization
 Speak to an Analyst
Speak to an Analyst
 Request Brochure
Request Brochure
 Inquire Before Buying
Inquire Before Buying




.webp)




.webp)












